

How Bubbles Form in Instrument Fluid Lines—and How Degassing Prevents Workflow Disruptions
How Bubbles Form in Instrument Fluid Lines
In liquid‑handling and analytical systems, dissolved gases can quietly undermine instrument precision, reproducibility, and detection accuracy. The series of phases below illustrates how bubbles form as fluid moves through a system—and why integrating a degasser before the flow path is the most effective way to eliminate these issues.
By conditioning the fluid before it enters the instrument, degassers remove dissolved gases at the source, reducing the risk of bubbles forming mid‑workflow and disrupting system performance.
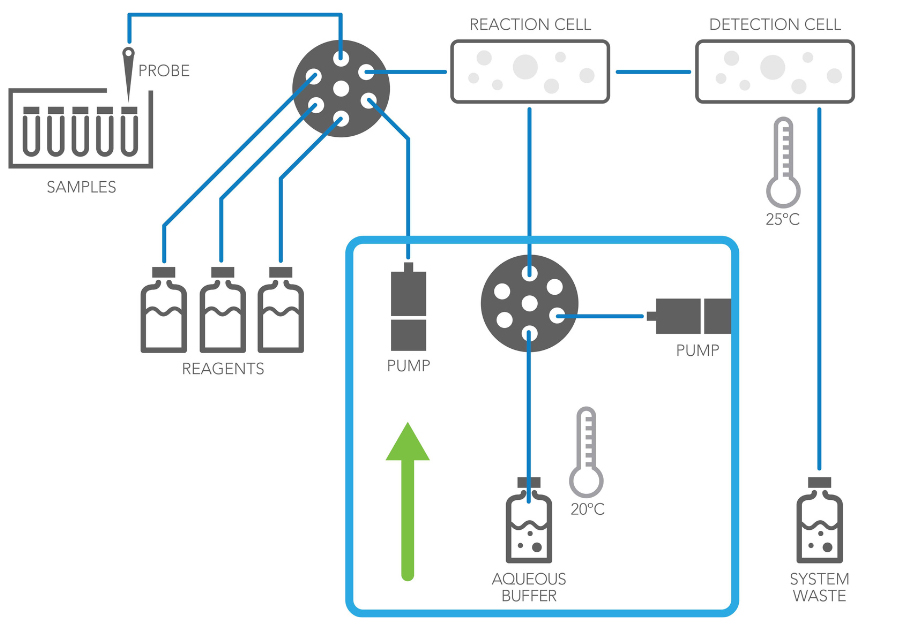
Phase 1: Pump Pulls Fluid into the System
When fluid is pulled from the reagent bottle into the instrument, it undergoes a transition from atmospheric pressure to system pressure.
At atmospheric pressure and 20°C, the solubility of air in water is 9.1 mg/L. As the pump draws fluid into the system (shown in the original green‑arrow graphic), pressure changes cause dissolved gases to escape in the form of bubbles within the tubing.
These bubbles can significantly affect pumping accuracy, system stability, and downstream performance.
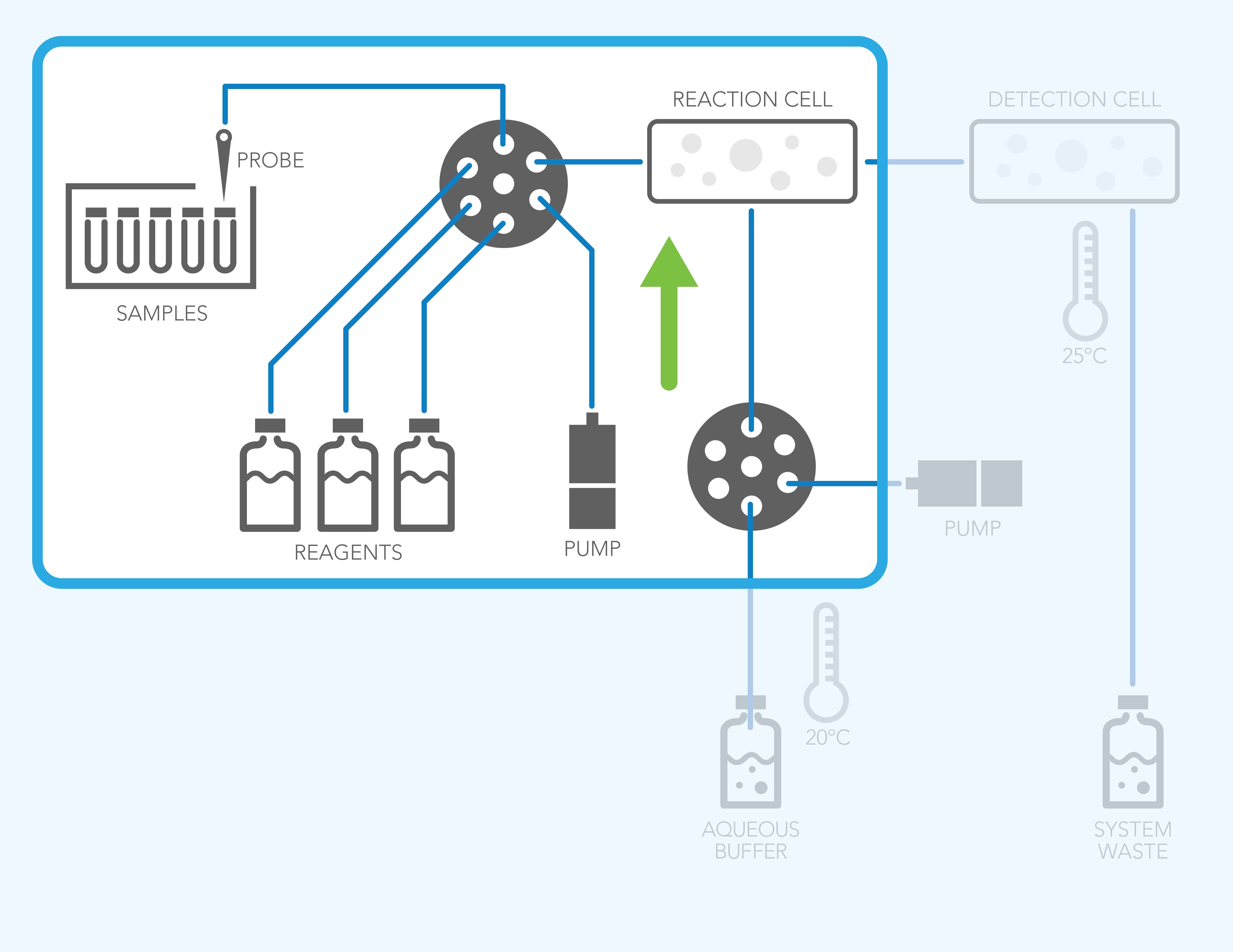
Phase 2: Mixing Reagents Can Lower Gas Solubility
Introducing samples, reagents, or solvents into the flow path changes system pressure, which can trigger bubble formation. Rapid temperature changes—especially from heating instruments—also reduce gas solubility.
When the fluid can no longer retain excess dissolved gas, bubbles outgas into the flow path, potentially impacting:
- Pumping stability
- Sample reproducibility
- Detector performance
- Overall system reliability
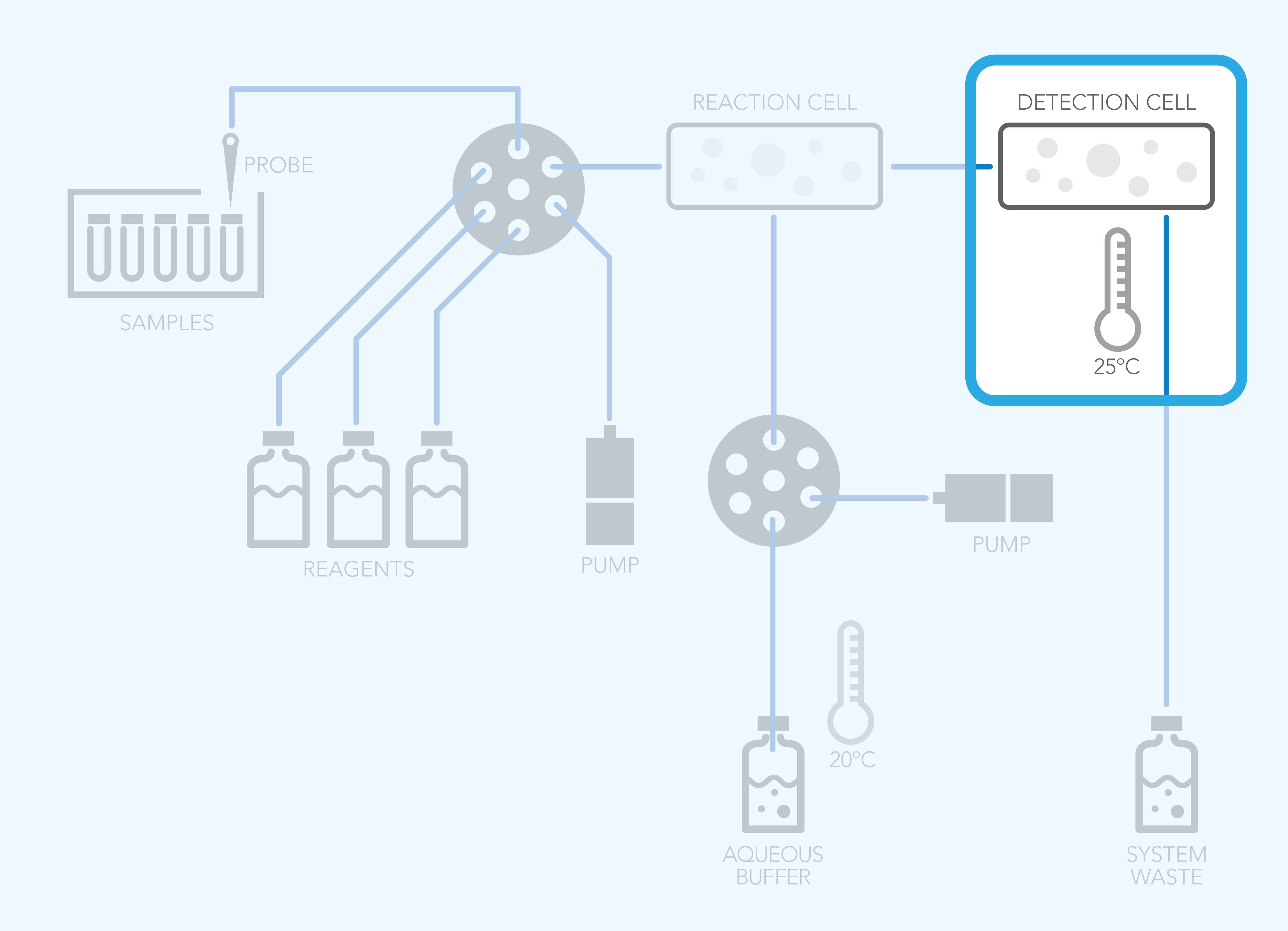
Phase 3: Detection and Heat Sources Can Increase Temperature
The detector cell is another high‑risk zone for pressure or temperature fluctuation.
Outgassed bubbles in the detector can cause:
- Sharp signal spikes
- Ghost peaks
- Elevated background noise
- Complete loss of detection
These issues often mimic “instrument failure,” when the real culprit is dissolved gas outgassing in the flow path.
Why Consider Degassing for Your Platform?
Dissolved gases are a hidden yet common source of issues in analytical instruments. Variations in temperature, pressure, and reagent composition all influence a fluid’s gas solubility—sometimes dramatically.
According to Henry’s Law, as temperature rises or pressure decreases, dissolved gases become less soluble and escape as bubbles.
These bubbles can:
- Disrupt pumping precision
- Cause noisy baselines
- Interfere with detector accuracy
- Create reproducibility challenges
- Lead to difficult installation troubleshooting—especially across diverse environmental conditions
Integrating a degasser reduces dissolved gas concentration below the saturation point, preventing outgassing throughout the entire system.
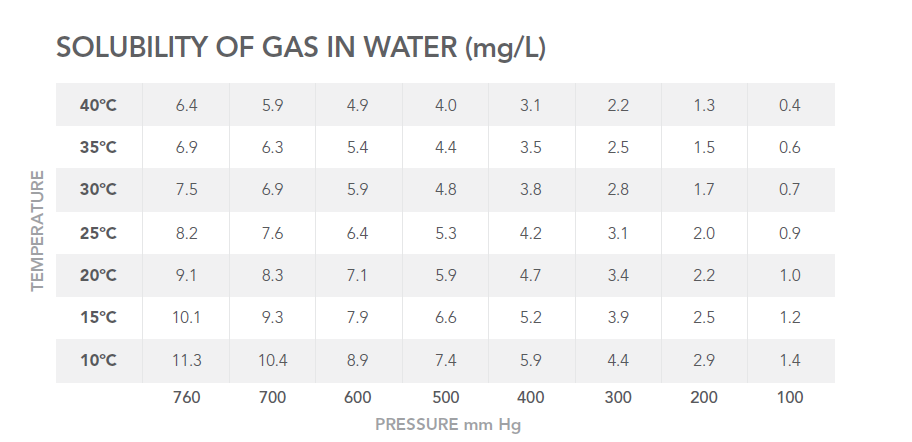
The Impact of Temperature and Pressure on System Fluids
The chart referenced in your original content illustrates how temperature and pressure affect the solubility of dissolved air in water.
Because dissolved gases can outgas at multiple points in an analytical workflow, they pose challenges for all liquid chemistries. Without degassing, even small fluctuations can lead to reliability and performance issues.
Why Partner with IDEX Health & Science for Your Degassing Needs?

We specialize in designing and manufacturing custom degassers tailored to meet the unique requirements of your application. With over 50 years of experience, our team of experts combines cutting-edge technology with unparalleled craftsmanship to deliver solutions that enhance efficiency, reliability, and performance. Our solutions can work with your on-board vacuum pump, or with our proven vacuum solutions. In addition to offering standalone degassing solutions, IDEX Health & Science can seamlessly integrate degassers onto manifolds for an elegant system design.

